PharmWare MCP Server for Claude Code 12 tools — connect in under 2 minutes
Claude Code is Anthropic's agentic CLI for terminal-first development. Add PharmWare as an MCP server in one command and Claude Code will discover every tool at runtime. ideal for automation pipelines, CI/CD integration, and headless workflows via Vinkius.
ASK AI ABOUT THIS MCP SERVER
Vinkius supports streamable HTTP and SSE.
Vinkius Desktop App
The modern way to manage MCP Servers — no config files, no terminal commands. Install PharmWare and 2,500+ MCP Servers from a single visual interface.
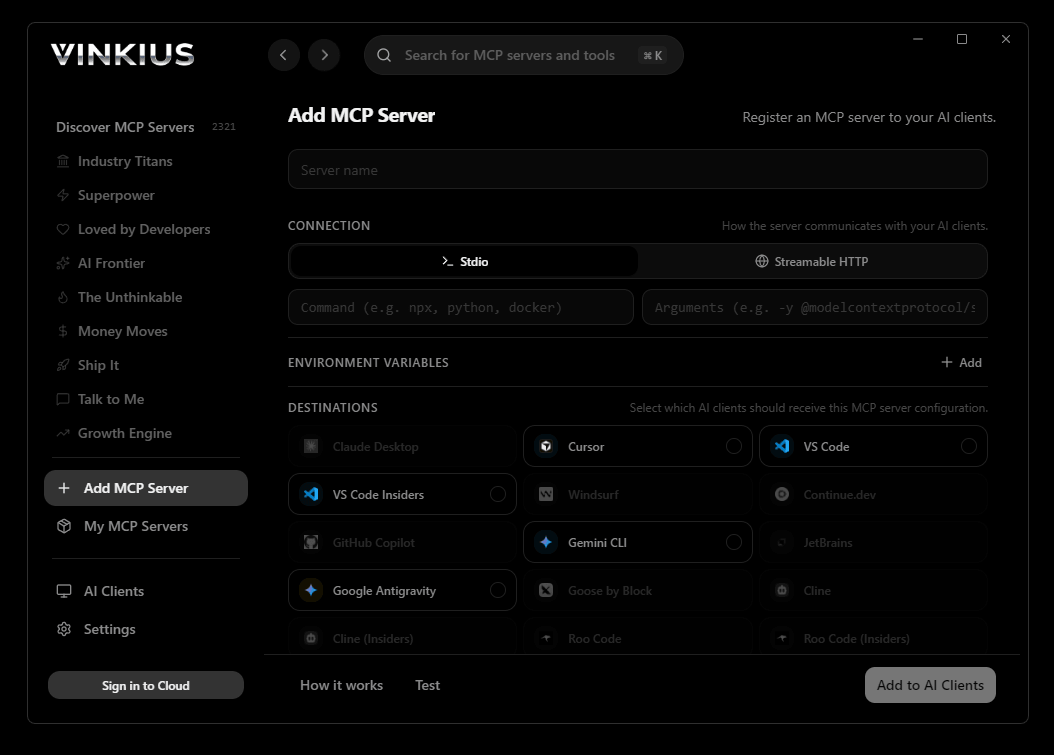

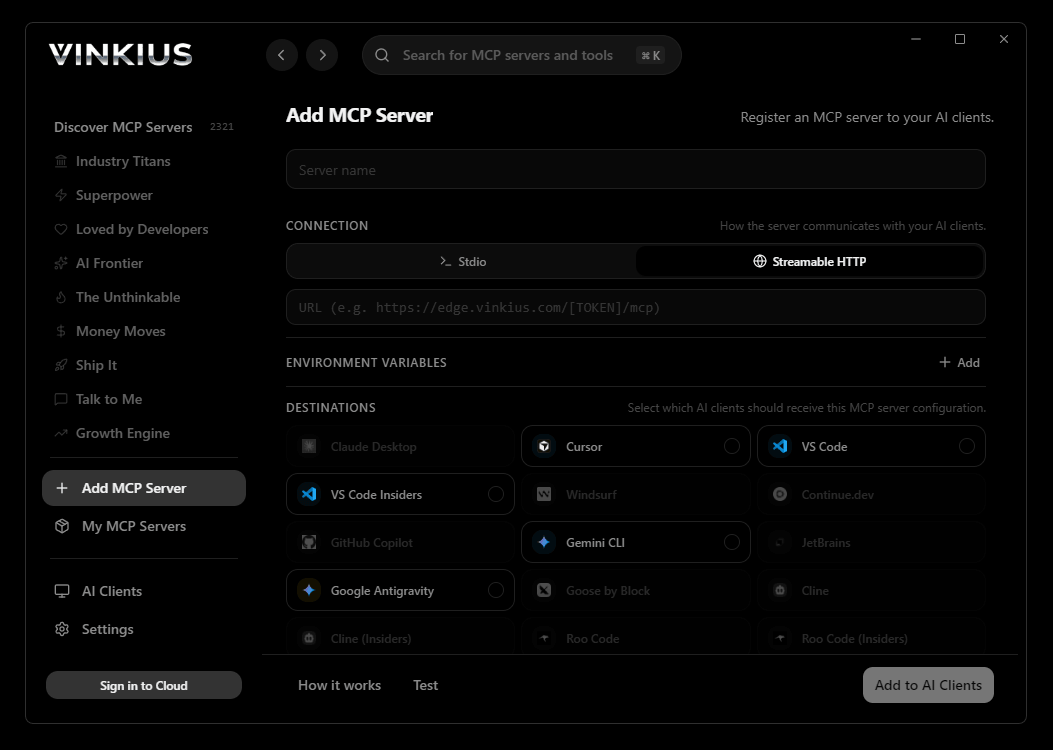
# Your Vinkius token. get it at cloud.vinkius.com
claude mcp add pharmware --transport http "https://edge.vinkius.com/[YOUR_TOKEN_HERE]/mcp"
* Every MCP server runs on Vinkius-managed infrastructure inside AWS - a purpose-built runtime with per-request V8 isolates, Ed25519 signed audit chains, and sub-40ms cold starts optimized for native MCP execution. See our infrastructure
About PharmWare MCP Server
Connect your PharmWare Cannabis Testing Laboratory Platform to any AI agent and take full control of your laboratory operations, quality assurance, and compliance workflows through natural conversation.
Claude Code registers PharmWare as an MCP server in a single terminal command. Once connected, Claude Code discovers all 12 tools at runtime and can call them headlessly. ideal for CI/CD pipelines, cron jobs, and automated workflows where PharmWare data drives decisions without human intervention.
What you can do
- Client Management — List all cultivators, processors, and retailers who submit samples to your laboratory for testing
- Sample Tracking — Monitor all cannabis samples received with chain of custody, testing status, and priority levels
- Test Panels — Browse available analytical methods (potency, terpenes, pesticides, heavy metals, microbials, mycotoxins)
- Test Results — Access complete analytical findings with pass/fail determinations against regulatory limits
- Certificates of Analysis — Retrieve all issued CoAs with QR codes for consumer verification and regulatory compliance
- Batch Traceability — Track production batches through laboratory testing with seed-to-sale linkage
- Laboratory Workflows — Monitor active processes from sample intake through CoA issuance with QC checkpoints
- Instrument Management — Verify calibration status and maintenance schedules for HPLC, GC-MS, ICP-MS systems
- Compliance Reports — Generate regulatory submissions, monthly summaries, and trend analyses
- Platform Integrations — Check health of connections to WeedMaps, Metrc, BioTrack, and state regulatory APIs
- User Administration — Review laboratory staff roles, permissions, and training certifications
- Audit Trails — Access complete operation logs for FDA 21 CFR Part 11 compliance and inspection readiness
The PharmWare MCP Server exposes 12 tools through the Vinkius. Connect it to Claude Code in under two minutes — no API keys to rotate, no infrastructure to provision, no vendor lock-in. Your configuration, your data, your control.
How to Connect PharmWare to Claude Code via MCP
Follow these steps to integrate the PharmWare MCP Server with Claude Code.
Install Claude Code
Run npm install -g @anthropic-ai/claude-code if not already installed
Add the MCP Server
Run the command above in your terminal
Verify the connection
Run claude mcp to list connected servers, or type /mcp inside a session
Start using PharmWare
Ask Claude: "Using PharmWare, show me...". 12 tools are ready
Why Use Claude Code with the PharmWare MCP Server
Claude Code provides unique advantages when paired with PharmWare through the Model Context Protocol.
Single-command setup: `claude mcp add` registers the server instantly. no config files to edit or applications to restart
Terminal-native workflow means MCP tools integrate seamlessly into shell scripts, CI/CD pipelines, and automated DevOps tasks
Claude Code runs headlessly, enabling unattended batch processing using PharmWare tools in cron jobs or deployment scripts
Built by the same team that created the MCP protocol, ensuring first-class compatibility and the fastest adoption of new protocol features
PharmWare + Claude Code Use Cases
Practical scenarios where Claude Code combined with the PharmWare MCP Server delivers measurable value.
CI/CD integration: embed PharmWare tool calls in your deployment pipeline to validate configurations or fetch secrets before shipping
Headless batch processing: schedule Claude Code to query PharmWare nightly and generate reports without human intervention
Shell scripting: pipe PharmWare outputs into other CLI tools for data transformation, filtering, and aggregation
Infrastructure monitoring: run Claude Code in a cron job to query PharmWare status endpoints and alert on anomalies
PharmWare MCP Tools for Claude Code (12)
These 12 tools become available when you connect PharmWare to Claude Code via MCP:
list_audit_logs
Each audit log entry contains the timestamp, performing user, action type (sample created, result modified, CoA issued, workflow completed, user permission changed), affected record ID, previous and new values (for modifications), IP address, and justification comment (if required). Fundamental for regulatory inspections, data integrity investigations, deviation root cause analysis, and FDA 21 CFR Part 11 compliance. AI agents use this to reconstruct event sequences during quality investigations, identify unauthorized changes, and generate audit-ready documentation packages. List all audit trail entries for laboratory operations and data changes
list_batches
Each batch entry contains the batch ID, producing facility license number, batch size, cannabis product type, harvest or manufacture date, linked samples submitted for testing, batch testing status (pending, partial, complete), disposition (released, quarantined, rejected, destroyed), and seed-to-sale tracking identifiers. Essential for batch-level compliance monitoring, recall management, and regulatory reporting. AI agents reference this when tracing contamination issues, verifying batch clearance for distribution, or generating lot-based compliance reports. List all cannabis batches tracked through laboratory testing
list_certificates
Each CoA record includes the certificate number, linked sample and batch, issuing laboratory accreditation details, comprehensive analytical results (potency profile, terpene fingerprint, contaminant screening), regulatory compliance statement, authorized signatory, issuance date, and QR code for consumer verification. Critical for product release decisions, regulatory audits, and consumer transparency programs. AI agents use this to verify CoA authenticity, batch compliance status, and generate client-facing documentation packages. List all Certificates of Analysis (CoA) issued by the laboratory
list_clients
Each client record contains company name, license number, contact information, client type (cultivation facility, processing plant, dispensary, or third-party tester), account status, and billing information. Essential for laboratory client management, sample intake workflows, and regulatory compliance reporting. AI agents should reference this when identifying sample ownership, generating client-specific reports, or verifying active testing contracts. List all clients (cultivators, processors, retailers) registered in PharmWare
list_instruments
Each instrument record contains the instrument name (HPLC system, GC-MS, ICP-MS, spectrophotometer), manufacturer, model, serial number, installation location, calibration status, last calibration date, next scheduled maintenance, qualification status (IQ/OQ/PQ), and associated test methods. Critical for instrument qualification, preventive maintenance scheduling, and analytical data integrity. AI agents should reference this to verify instrument readiness before assigning tests, schedule calibration activities, or troubleshoot analytical failures. List all laboratory instruments and equipment with calibration status
list_integrations
Each integration record contains the platform name (WeedMaps, Metrc, BioTrack, Leaf Data Systems, state regulatory API), integration type (bidirectional data sync, CoA publishing, sample status updates, regulatory reporting), connection status, last synchronization timestamp, data mapping configuration, and error logs. Critical for multi-platform compliance, automated CoA distribution, and real-time regulatory reporting. AI agents reference this to verify integration health, troubleshoot sync failures, and ensure seamless data flow between laboratory systems and external platforms. List all external system integrations (WeedMaps, state APIs, seed-to-sale platforms)
list_reports
Each report entry includes the report type (monthly summary, regulatory submission, client statement, trend analysis, deviation investigation, corrective action report), generation date, reporting period, associated clients or samples, regulatory agency destination (if applicable), and distribution status. Essential for regulatory compliance documentation, client billing reconciliation, and laboratory performance analytics. AI agents use this to prepare state-mandated reports, analyze testing trends, and identify quality improvement opportunities. List all laboratory reports and compliance documents generated
list_results
Each result contains the result ID, linked sample, test panel performed, analytical findings (THC/CBD potency percentages, terpene concentrations, pesticide residue levels, heavy metal concentrations, mycotoxin detection), pass/fail determination against regulatory limits, analyst who performed the test, review status, and date of completion. Fundamental for quality assurance, client notification workflows, and regulatory data submissions. AI agents should query this to verify sample compliance before releasing Certificates of Analysis or advising clients on product disposition. List all laboratory test results with analytical data
list_samples
Each sample contains the unique sample ID, submitting client, sample type (flower, edible, concentrate, topical, cartridge), received date, testing priority (standard, rush, priority), sample condition upon receipt, chain of custody documentation, and current testing status (received, in-progress, completed, failed). Critical for laboratory workflow management, turnaround time tracking, and seed-to-sale traceability compliance. AI agents use this to monitor sample queues, predict completion dates, and alert clients about status changes. List all cannabis samples submitted for laboratory testing
list_tests
Each test entry includes the test name (potency, terpenes, pesticides, heavy metals, mycotoxins, microbials, residual solvents, water activity, moisture content), test method (HPLC, GC-MS, ICP-MS, ELISA, qPCR), accreditation status, turnaround time, pricing, and regulatory limits per jurisdiction. Essential for test panel configuration, method validation, and compliance with state-specific cannabis testing requirements. AI agents reference this when configuring sample test orders, explaining testing scopes to clients, or verifying analytical method accreditation. List all test panels and analytical methods available in the laboratory
list_users
Each user record contains the username, full name, assigned role (laboratory director, quality manager, analytical chemist, sample technician, administrative staff), department, permission level (read-only, data entry, review/approval, system administrator), account status (active, inactive, locked), last login date, and training certification expiry. Essential for access control management, audit trail integrity, and ISO/IEC 17025 personnel competency requirements. AI agents should query this to verify user authorization before approving test results, assigning quality-critical tasks, or conducting access reviews. List all laboratory users with roles and permissions
list_workflows
Each workflow entry includes the workflow name (sample intake, potency testing, contaminant screening, CoA review, sample disposal), step definitions, assigned roles and responsibilities, quality control checkpoints, average completion time, and current instances in progress. Essential for laboratory operations management, staff task assignment, and process optimization. AI agents use this to guide technicians through testing procedures, identify workflow bottlenecks, and ensure ISO/IEC 17025 quality management system compliance. List all laboratory workflow templates and active processes
Example Prompts for PharmWare in Claude Code
Ready-to-use prompts you can give your Claude Code agent to start working with PharmWare immediately.
"Show me all cannabis samples currently in testing and their expected completion dates."
"List all pending Certificates of Analysis awaiting review and signature."
"Check the integration status with state regulatory APIs and WeedMaps CoA publishing."
Troubleshooting PharmWare MCP Server with Claude Code
Common issues when connecting PharmWare to Claude Code through the Vinkius, and how to resolve them.
Command not found: claude
npm install -g @anthropic-ai/claude-codeConnection timeout
PharmWare + Claude Code FAQ
Common questions about integrating PharmWare MCP Server with Claude Code.
How do I add an MCP server to Claude Code?
claude mcp add --transport http "" in your terminal. Claude Code registers the server and discovers all tools immediately.Can Claude Code run MCP tools in headless mode?
How do I list all connected MCP servers?
claude mcp in your terminal to see all registered servers and their status, or type /mcp inside an active Claude Code session.Connect PharmWare with your favorite client
Step-by-step setup guides for every MCP-compatible client and framework:
Anthropic's native desktop app for Claude with built-in MCP support.
AI-first code editor with integrated LLM-powered coding assistance.
GitHub Copilot in VS Code with Agent mode and MCP support.
Purpose-built IDE for agentic AI coding workflows.
Autonomous AI coding agent that runs inside VS Code.
Anthropic's agentic CLI for terminal-first development.
Python SDK for building production-grade OpenAI agent workflows.
Google's framework for building production AI agents.
Type-safe agent development for Python with first-class MCP support.
TypeScript toolkit for building AI-powered web applications.
TypeScript-native agent framework for modern web stacks.
Python framework for orchestrating collaborative AI agent crews.
Leading Python framework for composable LLM applications.
Data-aware AI agent framework for structured and unstructured sources.
Microsoft's framework for multi-agent collaborative conversations.
Connect PharmWare to Claude Code
Get your token, paste the configuration, and start using 12 tools in under 2 minutes. No API key management needed.
